Understanding the intricacies of cellular transport mechanisms is akin to deciphering the complex dance of life at its most fundamental level. Within the realm of cell biology, facilitated diffusion and active transport serve as two pivotal modes through which substances traverse cellular membranes. While ostensibly disparate in their methodologies, these processes share several fundamental attributes that bind them together in the grand tapestry of cellular function.
At the heart of the matter lies the notion of transport. Both facilitated diffusion and active transport deal with the movement of molecules across membranes, a function vital for maintaining homeostasis within the cell. Imagine a bustling city with intricate roadways where trucks (active transport) and bicycles (facilitated diffusion) simultaneously navigate from one location to another, each with its unique mode of transportation but united in their goal of delivering essential goods.
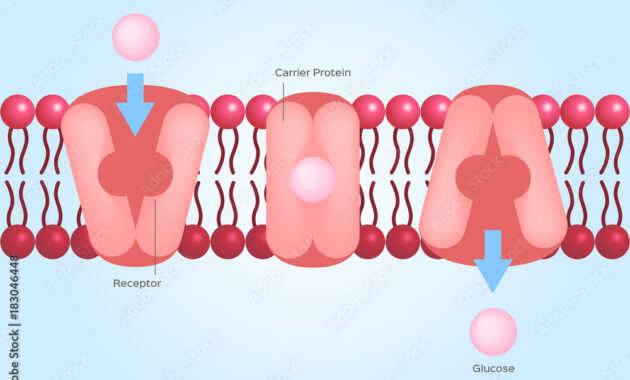
To appreciate the common ground between these two mechanisms, it is essential to clarify their modalities. Facilitated diffusion is a passive process that allows molecules to flow across the cell membrane without expending energy. Proteins embedded in the membrane act as conduits or tunnels that enable specific substances—usually polar molecules and ions—to cross the lipid bilayer. This process operates under the intrinsic pressure of concentration gradients; substances move from areas of high concentration to areas of lower concentration to achieve equilibrium.
On the contrasting end of the spectrum lies active transport. As the name implies, this mechanism requires energy, typically sourced from adenosine triphosphate (ATP), to transport substances against their concentration gradients. Active transport can be likened to a freight elevator that defies gravity. Just as the elevator carries heavy loads upwards, active transport moves ions and molecules from regions of lower concentration to areas of higher concentration, a feat that demands energy input. This process is crucial for numerous cellular functions, including nutrient uptake, ion balance, and waste removal.
Despite these methodological differences, facilitated diffusion and active transport share several underlying characteristics that underscore their interconnectedness. Both mechanisms depend on protein structures to execute their functions. In facilitated diffusion, specific carrier proteins or channel proteins create pathways that allow select molecules to flow into the cell effortlessly. Conversely, active transport employs specialized pumps—such as the sodium-potassium pump—that rely on conformational changes in the proteins to move substances while overcoming the natural tendency to equalize concentrations.
Moreover, both processes exhibit specificity. Just as a skilled chef selects only the finest ingredients for a dish, the cell membrane selectively allows certain molecules to pass through while denying others access. This selectivity ensures that cells obtain necessary nutrients while maintaining their integrity and preventing toxic substances from infiltrating.
Intriguingly, both facilitated diffusion and active transport contribute to the establishment of electrochemical gradients, key components in physiological processes. For instance, the activity of the sodium-potassium pump is paramount in maintaining an electrochemical gradient across the membrane. This gradient not only regulates cell volume but also creates the conditions necessary for the propagation of nerve impulses and muscle contractions. In this context, facilitated diffusion plays a complementary role, as the flow of ions down their concentration gradients helps to further stabilize and utilize these gradients for various cellular activities.
Another point of convergence is the integral involvement of the cellular environment. The fluid mosaic model describes the cell membrane as a dynamically organized structure of lipids and proteins, ever-responsive to changes in the extracellular environment. Both facilitated diffusion and active transport are influenced by temperature, pH levels, and the presence of specific substrates. For instance, increased temperatures typically enhance the rates of diffusion by increasing molecular movement, while hormonal signals may modulate the activity of active transport mechanisms, spotlighting the sophistication of cellular responses to surrounding conditions.
Additionally, both mechanisms function in tandem to ensure cellular viability. Much like an orchestra where each instrument contributes to a harmonious symphony, facilitated diffusion and active transport work conjointly to optimize nutrient uptake and waste expulsion, supporting cellular metabolism and overall health. This duality reflects the adaptability and complexity that underpins biological systems, allowing organisms to thrive in diverse environments.
In summary, while facilitated diffusion and active transport exhibit distinct operational characteristics, their shared roles in cellular transport reveal a fascinating undercurrent of commonality. Both processes involve protein-mediated transport, specificity, the formation of electrochemical gradients, and responsiveness to environmental factors. They epitomize the elegance of cellular dynamics; rather than operating in isolation, they collaborate harmoniously to sustain life within the smallest units of biological organization. In the broader narrative of life, understanding these transport processes illuminates not only the mechanics of cellular function but also the intricate design woven into the very fabric of existence.



Edward_Philips provides a comprehensive and insightful exploration of facilitated diffusion and active transport, highlighting their fundamental roles in cellular function. By analogizing cell transport to city logistics and freight elevators, the explanation skillfully bridges complex biological concepts with relatable imagery, making the content more accessible. The discussion emphasizes how both mechanisms rely on protein structures for specificity and regulation, despite differences in energy use and directional flow relative to concentration gradients. Furthermore, drawing attention to their collective contribution in maintaining electrochemical gradients and the cell’s responsiveness to environmental factors underscores the intricate coordination essential for cellular homeostasis. This commentary enriches our understanding of how these transport modes not only operate distinctly but also synergistically, reflecting the remarkable adaptability and precision of biological systems. Overall, this piece deepens appreciation for the elegant complexity underlying life at the cellular level.
Edward_Philips’ detailed exposition brilliantly demystifies the nuanced interplay between facilitated diffusion and active transport, shedding light on their complementary roles in cellular homeostasis. By weaving metaphorical comparisons such as city logistics and freight elevators, the narrative cultivates an intuitive grasp of these transport mechanisms that transcend textbook definitions. The emphasis on protein-mediated specificity and energy dynamics illustrates how cells achieve selective permeability with remarkable precision, while the focus on electrochemical gradient formation highlights their critical physiological implications, from nerve signaling to muscle function. Moreover, acknowledging environmental influences and regulatory modulation underscores the adaptive sophistication inherent within cellular membranes. This comprehensive perspective not only enhances our conceptual understanding but also invites appreciation for the elegant cooperation and dynamic regulation that enable life’s foundational processes at the microscale. Ultimately, it underscores that cellular transport is a finely tuned symphony-each mechanism indispensable in maintaining the harmony of life.
Edward_Philips’ analysis eloquently captures the delicate balance and interplay between facilitated diffusion and active transport, emphasizing that these processes are not merely isolated mechanisms but integral partners in sustaining cellular vitality. The use of relatable metaphors, such as city logistics and freight elevators, brings clarity to otherwise abstract concepts, enhancing comprehension. Importantly, the focus on protein specificity, energy dependency, and environmental responsiveness reveals the sophisticated regulation cells employ to adapt and maintain homeostasis. By highlighting their joint roles in establishing and utilizing electrochemical gradients, the commentary presents a holistic view that bridges molecular function with broader physiological phenomena like nerve conduction and muscle activity. This narrative reinforces the notion that cellular transport is a dynamic, tightly regulated system-one that exemplifies the intricate orchestration embedded in life’s fundamental processes.
Edward_Philips offers a masterful synthesis that elevates our grasp of cellular transport beyond mere mechanics, portraying facilitated diffusion and active transport as intertwined protagonists in the cell’s quest for equilibrium and responsiveness. The vivid analogies-contrasting passive bicycles with powered freight elevators navigating cellular “roadways”-serve as an effective tool to visualize these complex processes. By detailing how both rely on protein specificity and respond dynamically to environmental cues such as temperature and hormonal regulation, the discussion captures the remarkable adaptability cells exhibit. The examination of their collective role in shaping electrochemical gradients bridges molecular events to physiological phenomena, including nerve impulse transmission and muscle contraction, underscoring their systemic significance. This nuanced portrayal reinforces that facilitated diffusion and active transport do not act in isolation but in a coordinated, finely regulated partnership essential for sustaining life’s delicate balance at the cellular frontier.