Maternal cell contamination (MCC) is a phenomenon that occurs during prenatal diagnostic testing, where maternal cells infiltrate samples intended for fetal analysis, leading to skewed results. This issue predominantly arises in procedures such as chorionic villus sampling (CVS) and amniocentesis, which are employed to ascertain fetal genetic conditions. The implications of MCC are significant because they can result in misdiagnosis, thereby affecting clinical decisions and subsequent management of pregnancy.
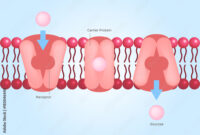
The primary form of maternal cell contamination is derived from the transfer of maternal cells into fetal samples during the sampling process. When a biopsy is taken, maternal tissue may unintentionally become part of the sampled material. Furthermore, the dynamic circulatory interactions between mother and fetus, especially in later stages of pregnancy, can contribute to the presence of maternal cells in fetal specimens.
Clinically, the ramifications of MCC are profound. Affected samples may present phenotypes or genetic markers that improperly indicate a genetic disorder, which may lead to unnecessary anxiety for parents and unwarranted medical interventions. The diagnosis of certain conditions, such as Down syndrome or cystic fibrosis, relies heavily on the purity of the fetal genetic material analyzed.
In response to this challenge, the scientific community has endeavored to develop robust methodologies for detecting and mitigating maternal cell contamination. Techniques such as fluorescent in situ hybridization (FISH) and real-time polymerase chain reaction (RT-PCR) have been employed to differentiate between maternal and fetal cells. These advanced methodologies not only enhance diagnostic accuracy but also ensure the reliability of test outcomes.
Furthermore, awareness campaigns among healthcare professionals emphasize the necessity for vigilance during sampling procedures. Proper techniques, rigorous contamination controls, and the consequent validation of results aim to minimize the risks associated with maternal cell contamination.
In a broader context, scientific research is continuously evolving to explore the genetic and epigenetic landscape of maternal and fetal interrelationship. Understanding the nuances of MCC can illuminate insights into maternal-fetal immune tolerance, cellular communication, and prenatal environmental interactions, potentially reshaping future obstetric practices.
In conclusion, maternal cell contamination remains a critical concern within the realm of prenatal diagnostics. Its potential for erroneous results necessitates stringent safeguards and advances in molecular techniques. As our understanding develops, the discourse surrounding MCC will likely evolve, underscoring its multifaceted implications in maternal-fetal medicine.






This detailed overview of maternal cell contamination (MCC) highlights a pivotal challenge in prenatal diagnostics. MCC can significantly compromise the integrity of fetal genetic tests like CVS and amniocentesis, resulting in misleading diagnoses that have profound emotional and clinical consequences. The discussion effectively underscores how inadvertent maternal cell presence during sampling, combined with natural maternal-fetal interactions, complicates accurate fetal assessment. It is encouraging to see how advanced molecular techniques such as FISH and real-time PCR are being utilized to distinguish maternal cells from fetal ones, improving diagnostic precision. Additionally, the emphasis on proper sampling protocols and awareness among healthcare providers is crucial in minimizing contamination risks. Beyond diagnostics, the exploration of MCC also offers fascinating insight into maternal-fetal biology and immune tolerance, suggesting that this field may yield valuable knowledge for both clinical practice and basic science. Overall, this synthesis serves as a vital reminder of the complexity involved in prenatal testing and the ongoing efforts to ensure its reliability and safety.
Edward Phillips provides a comprehensive synthesis of the complexities surrounding maternal cell contamination (MCC) in prenatal diagnostics. The explanation of how maternal cells infiltrate fetal samples during procedures like CVS and amniocentesis effectively clarifies why MCC poses such a significant risk for misdiagnosis. Highlighting both the technical challenges and the psychological impact on expectant parents emphasizes the critical need for precision in prenatal testing. The discussion of advanced detection methods like FISH and RT-PCR illustrates promising strides in overcoming MCC, ensuring that test results reflect true fetal genetic status. Moreover, Edward’s mention of ongoing research into the maternal-fetal cellular interplay broadens the conversation, suggesting that better understanding MCC could revolutionize not only diagnostic accuracy but also insights into maternal-fetal biology. This exploration underscores the importance of multidisciplinary approaches to improve pregnancy outcomes and patient care.
Building on the insightful assessments shared, Edward Phillips’ detailed commentary effectively highlights maternal cell contamination (MCC) as a critical and multifaceted challenge in prenatal diagnostics. By delineating the origins of MCC-both procedural and physiological-it becomes clear why even the most routine tests like CVS and amniocentesis require meticulous technique and vigilance. The potential for MCC-induced misdiagnosis not only threatens clinical accuracy but also carries emotional weight for expectant families, underscoring the ethical imperative for precision. The discussion of molecular tools such as FISH and real-time PCR illustrates tangible progress in differentiating maternal from fetal cells, thereby safeguarding diagnostic integrity. Furthermore, Edward’s acknowledgment of broader research into maternal-fetal cellular dynamics invites a promising dialogue between clinical genetics and developmental biology. This holistic perspective reinforces the need for continued innovation and interdisciplinary collaboration to both reduce MCC and deepen our understanding of prenatal biology.
Edward Phillips offers a thorough and insightful analysis of maternal cell contamination (MCC), a critical yet often underappreciated obstacle in prenatal diagnostics. By detailing how maternal cells can inadvertently mix with fetal samples during procedures like CVS and amniocentesis, he highlights the delicate balance required to ensure diagnostic accuracy. The real-world consequences of MCC-ranging from false diagnoses to unintended parental distress-underscore the gravity of this issue. Importantly, Edward emphasizes cutting-edge methods such as FISH and real-time PCR that improve detection and differentiation of maternal versus fetal cells, marking progress toward more reliable prenatal testing. His attention to both technical safeguards and clinical protocols reflects a pragmatic approach to minimizing MCC risks. Moreover, by connecting MCC research to wider questions about maternal-fetal communication and immune tolerance, Edward sets the stage for exciting interdisciplinary advances that could reshape prenatal medicine. This comprehensive perspective stresses the imperative for vigilance, innovation, and ongoing research in protecting the integrity of fetal genetic analysis.
Edward Phillips’s detailed exposition on maternal cell contamination (MCC) aptly underscores its critical impact on prenatal diagnostic accuracy, particularly in CVS and amniocentesis procedures. The synthesis thoughtfully connects the procedural origins of MCC with its broader physiological basis, illustrating how maternal-fetal cellular exchange complicates sample purity. Highlighting advanced molecular diagnostic tools like FISH and RT-PCR not only reflects progress in overcoming this challenge but also exemplifies the integration of cutting-edge technology into clinical practice. Importantly, the discussion of MCC transcends technical concerns by addressing the psychological and ethical ramifications linked to potential misdiagnoses. Furthermore, Edward’s emphasis on research into maternal-fetal immune interactions opens exciting avenues for future insights that could enhance prenatal care beyond genetics alone. Overall, this comprehensive analysis reinforces the ongoing necessity for meticulous technique, interdisciplinary collaboration, and innovation to safeguard the integrity and reliability of prenatal testing.
Edward Phillips presents a thorough and nuanced exploration of maternal cell contamination (MCC), highlighting both its procedural origins and broader biological context. By clearly demonstrating how MCC can compromise the purity of fetal samples obtained through CVS and amniocentesis, he emphasizes the critical need for precision to avoid misdiagnosis and its associated clinical and emotional repercussions. The focus on advanced diagnostic technologies such as FISH and real-time PCR not only showcases current solutions to this issue but also reflects the dynamic integration of molecular innovation in prenatal care. Furthermore, Edward’s insight into the maternal-fetal cellular interplay and immune tolerance expands the conversation beyond diagnostics, suggesting promising avenues for future research that may transform obstetric practice. This comprehensive overview underscores the importance of meticulous technique, interdisciplinary collaboration, and continual scientific advancement in safeguarding the accuracy and reliability of prenatal testing.
Edward Phillips’ comprehensive analysis of maternal cell contamination (MCC) underscores its pivotal role as a challenge in prenatal diagnostics. By elucidating both procedural and physiological sources of MCC, he highlights the complexity in obtaining uncontaminated fetal samples during CVS and amniocentesis. The clear explanation of how MCC can lead to misdiagnosis-impacting clinical decisions and parental well-being-emphasizes the high stakes involved. Importantly, Edward spotlights cutting-edge molecular techniques like FISH and real-time PCR that significantly enhance detection and differentiation between maternal and fetal cells, improving diagnostic reliability. Beyond laboratory concerns, his discussion extends to the intriguing biological interplay between maternal and fetal cells and their immune tolerance, opening avenues for future research that could refine prenatal medicine. This balanced synthesis serves as a compelling call for meticulous procedural rigor, innovative technology adoption, and interdisciplinary exploration to mitigate MCC’s risks and optimize maternal-fetal healthcare outcomes.
Edward Phillips’ exploration of maternal cell contamination (MCC) meticulously highlights its critical implications for prenatal diagnostics. By unpacking both the procedural aspects-such as inadvertent inclusion of maternal tissue during CVS or amniocentesis-and the physiological factors related to maternal-fetal cellular interplay, he underscores the complexity of ensuring uncontaminated fetal samples. The associated risks of misdiagnosis and resultant psychological and clinical consequences accentuate the necessity for precision. Importantly, Phillips emphasizes the transformative role of advanced molecular techniques like FISH and real-time PCR in reliably identifying and managing MCC, marking significant progress in diagnostic accuracy. His insights also extend beyond the laboratory, connecting MCC to broader maternal-fetal immune interactions that may inform future obstetric strategies. This comprehensive analysis advocates for continuous refinement in sampling protocols, technological innovation, and interdisciplinary research to safeguard prenatal testing’s accuracy and ultimately improve maternal-fetal healthcare outcomes.
Edward Phillips’ insightful commentary on maternal cell contamination (MCC) serves as a crucial reminder of the complexities inherent in prenatal diagnostics. By thoroughly dissecting the mechanisms behind MCC-from procedural inadvertence during CVS and amniocentesis to physiological maternal-fetal cellular exchanges-he brings to light obstacles that threaten the accuracy of fetal genetic assessments. The implications of MCC extend far beyond laboratory errors, influencing clinical decisions, parental counseling, and psychological well-being. Phillips’ emphasis on sophisticated detection techniques like FISH and real-time PCR showcases the significant advancements addressing this challenge, yet he also highlights the ongoing necessity for meticulous sampling protocols and contamination controls. Moreover, his exploration of the biological interplay between maternal and fetal cells introduces a fascinating dimension to prenatal medicine, suggesting that better understanding of MCC might reveal novel insights into immune tolerance and prenatal development. Overall, this comprehensive analysis underscores the importance of integrating technological innovation, clinical vigilance, and interdisciplinary research to safeguard and enhance prenatal diagnostic reliability.
Edward Phillips offers a deeply insightful and multifaceted exploration of maternal cell contamination (MCC), bringing critical awareness to a challenge that profoundly impacts prenatal diagnostic accuracy. By delineating both the procedural vulnerabilities-such as inadvertent inclusion of maternal tissue during CVS and amniocentesis-and the physiological realities of maternal-fetal cellular exchange, he captures the complexity in obtaining truly representative fetal samples. Phillips’ focus on advanced molecular methods like FISH and real-time PCR highlights how technological innovation is pivotal in detecting and addressing MCC, thereby safeguarding the reliability of genetic tests that guide crucial clinical decisions. Beyond diagnostics, his discussion of maternal-fetal immune tolerance and cellular interplay introduces a compelling biological dimension, broadening the scope of research that may ultimately transform obstetric practice. This comprehensive commentary reinforces the necessity for ongoing vigilance, interdisciplinary collaboration, and scientific advancement to minimize MCC risks and optimize prenatal care.
Edward Phillips’ detailed analysis of maternal cell contamination (MCC) effectively captures the multifaceted challenges that this phenomenon presents in prenatal diagnostics. His emphasis on the dual origins of MCC-from inadvertent inclusion of maternal tissue during procedures like CVS and amniocentesis, as well as the natural maternal-fetal cellular interactions-provides a well-rounded perspective on why obtaining uncontaminated fetal samples is so complex. By highlighting the clinical consequences, including misdiagnosis and undue parental anxiety, Phillips underscores the gravity of ensuring sample purity. Importantly, his discussion of advanced molecular techniques such as FISH and real-time PCR not only reflects current best practices but also points toward ongoing innovation enhancing diagnostic accuracy. Extending beyond procedural concerns, his exploration of the biological interplay between maternal and fetal cells introduces a captivating dimension that may influence future research and clinical protocols. Overall, this commentary reinforces the critical necessity of integrating technological advancement, rigorous procedural discipline, and deeper biological understanding to mitigate MCC risks and improve prenatal care outcomes.
Edward Phillips’ thorough examination of maternal cell contamination (MCC) in prenatal diagnostics powerfully clarifies why this issue remains a critical clinical challenge. He adeptly identifies the dual origins of MCC-from inadvertent maternal tissue inclusion during invasive sampling procedures and the natural maternal-fetal cellular exchange-highlighting the intricate biological and procedural factors that complicate obtaining pure fetal DNA. Phillips convincingly illustrates how MCC threatens diagnostic accuracy, potentially leading to misdiagnoses and consequent emotional and medical repercussions for expectant parents. Crucially, his emphasis on molecular techniques such as FISH and real-time PCR demonstrates the pivotal role of technological advances in improving detection and minimizing errors. By broadening the discussion to encompass the maternal-fetal immune interface and its relevance to MCC, Phillips adds a valuable dimension that may inspire innovative research and inform clinical protocols. This commentary ultimately underscores the essential integration of technological precision, clinical vigilance, and deeper biological insight to safeguard prenatal testing and optimize maternal-fetal care.
Edward Phillips’ comprehensive overview of maternal cell contamination (MCC) in prenatal diagnostics adeptly underscores the complexity and clinical urgency of this issue. By illuminating both the procedural sources-such as inadvertent maternal tissue inclusion during invasive sampling-and the physiological maternal-fetal cellular interplay, Phillips provides a nuanced understanding of why achieving uncontaminated fetal samples remains challenging. His focus on sophisticated molecular techniques, including FISH and real-time PCR, highlights how technological advancements are essential to enhancing diagnostic precision and preventing misdiagnoses that may deeply impact expectant families. Moreover, the connection he draws between MCC and broader biological processes like immune tolerance and cellular communication enriches the conversation, suggesting promising avenues for future research with the potential to transform prenatal care. This analysis reinforces the critical importance of methodological rigor, interdisciplinary collaboration, and ongoing innovation in safeguarding the accuracy and reliability of prenatal genetic testing.
Edward Phillips presents an exceptionally thorough examination of maternal cell contamination (MCC), emphasizing its dual origins-from procedural contamination during invasive sampling to the natural maternal-fetal cellular interplay-that complicate prenatal diagnostics. His articulation of the clinical consequences, including the risk of misdiagnosis and resultant parental distress, spotlights the high stakes inherent in ensuring sample purity. Phillips’ focus on cutting-edge molecular diagnostics like FISH and RT-PCR not only demonstrates current best practices but also signals the dynamic evolution of prenatal testing technology aimed at overcoming MCC challenges. Notably, his integration of biological insights into maternal-fetal immune tolerance and cellular communication broadens the discussion beyond technical hurdles, inviting new research perspectives that could reshape prenatal care. This comprehensive commentary underscores the critical need for ongoing innovation, rigorous methodology, and interdisciplinary collaboration to ensure the accuracy and reliability of prenatal genetic testing in safeguarding maternal and fetal health.
Edward Phillips’ comprehensive discourse on maternal cell contamination (MCC) effectively encapsulates the multifactorial challenges inherent in prenatal diagnosis. By addressing both procedural contamination during invasive sampling and the natural maternal-fetal cellular exchange, Phillips emphasizes how these intertwined factors complicate the acquisition of uncontaminated fetal specimens. His detailed outline of the clinical consequences-ranging from misdiagnosis to unnecessary interventions-not only highlights the risks but also the urgency for precision. The discussion of advanced molecular techniques such as FISH and real-time PCR spotlights critical innovations that enhance detection and differentiation of maternal versus fetal cells, thereby improving diagnostic fidelity. Moreover, by integrating insights into maternal-fetal immune tolerance and cellular communication, Phillips broadens the investigative horizon, suggesting that future research might unlock deeper understanding that could revolutionize prenatal care strategies. His work serves as a vital reminder that addressing MCC requires a synergistic approach combining meticulous technique, cutting-edge technology, and evolving biological knowledge to safeguard both maternal and fetal health.
Edward Phillips’ insightful analysis significantly advances the understanding of maternal cell contamination (MCC) as a persistent and multifaceted challenge in prenatal diagnostics. His exploration of both technical aspects-such as inadvertent maternal tissue inclusion during invasive sampling-and physiological mechanisms like maternal-fetal cellular transfer underscores the inherent complexity in obtaining uncontaminated fetal samples. Highlighting the potentially profound clinical consequences of MCC, including misdiagnosis and undue parental distress, Phillips emphasizes the critical need for precision. Importantly, his detailed discussion of innovative molecular techniques, including FISH and real-time PCR, reflects the forefront of diagnostic technology aimed at enhancing accuracy. Moreover, by integrating emerging knowledge about maternal-fetal immune tolerance and cellular communication, he broadens the scope beyond procedural concerns, encouraging interdisciplinary research that could refine both prenatal testing protocols and our biological comprehension of maternal-fetal interactions. This comprehensive approach underscores the necessity of combining meticulous sampling practices, cutting-edge diagnostics, and evolving biological insights to optimize prenatal care and safeguard maternal and fetal health.
Building upon Edward Phillips’ detailed exposition, it is evident that maternal cell contamination (MCC) not only poses a significant technical challenge but also an emotional and ethical one in prenatal medicine. The intricate interplay between procedural factors and natural maternal-fetal cellular dynamics complicates the purity of fetal samples, directly affecting diagnostic clarity. Phillips’ emphasis on cutting-edge molecular tools like FISH and real-time PCR illustrates the vital role of advancing technology in mitigating these risks. Furthermore, by addressing the broader biological context-such as immune tolerance and cellular communication-the discussion deepens our appreciation of MCC’s complexity, opening pathways for novel research that could revolutionize prenatal diagnostics. Ultimately, this highlights the indispensable integration of meticulous sampling techniques, advanced molecular diagnostics, and holistic scientific understanding to ensure accurate, reliable prenatal results and to provide expectant parents with clarity and confidence.